111年:(醫檢)生化(2)
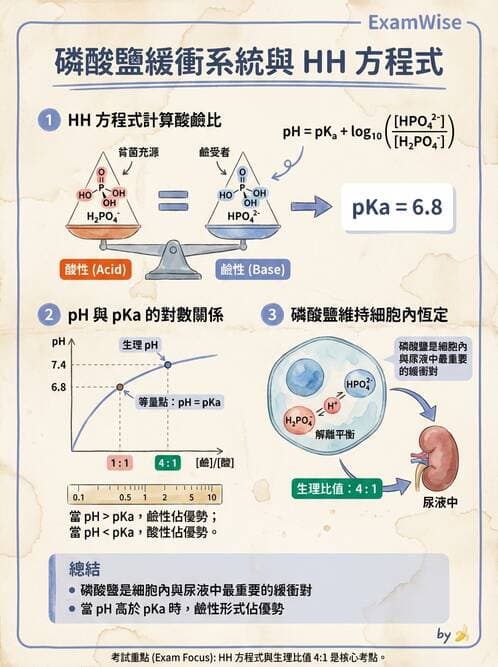
若病人血液 pH = 7.4 , [ 依據 Henderson-Hasselbalch equation 推算: pH = pKa+log( HPO₄²⁻/ H₂PO₄⁻), pKa =6.8],則此病人檢體中磷酸氫根( HPO₄²⁻)與磷酸二氫根( H₂PO₄⁻)之比值約為多少?
A1
B4
C10
D16
詳細解析
計算步驟如下
Henderson–Hasselbalch 方程式:
pH = pKa + log([HPO₄²⁻] / [H₂PO₄⁻]) (pearson.com)
-
已知 pH = 7.4,pKa = 6.8 (磷酸二氫根 H₂PO₄⁻ 的 pKa) (labtestsguide.com),帶入:
7.4 = 6.8 + log(ratio) -
解出 log(ratio) = 7.4 − 6.8 = 0.6,因此
ratio = 10⁰·⁶ ≈ 4.0 ([gauthmath.com](https://www.gauthmath.com/solution/1801455047140358/A-solution-containing-phosphate-buffer-pKa-6-8-has-a-pH-of-7-
...(解析預覽)...
升級 VIP 解鎖圖文解析